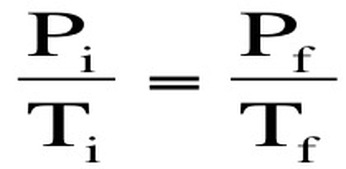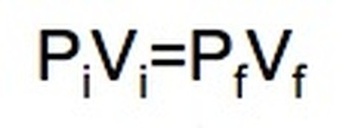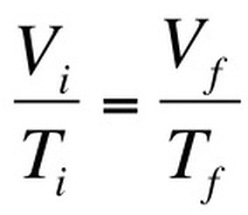- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 8 - Direct and Indirect Love of Chemistry
|
We all know and love the gas laws, but how do we know that these are true. The only way that we can know if something is true or not in science is through experimental evidence. Therefore, in this lab you will designing and implementing an experiment that provides evidence that supports that conclusions that are made from Boyle's, Charles', and Gay-Lussac's Law.
|
The Gas Laws
|
We can describe how a gas will change based on the volume, pressure, temperature, and moles of the gas. The gas laws focus on these properties and how changing one affects the other three.
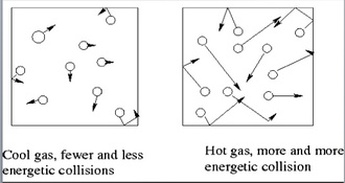
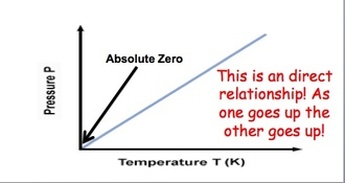
Gay Lussac's Law: Gas molecules move quicker the more we heat them up and increase the temperature. This means that they will be hitting the sides of the container more often and the pressure will also increase as the temperature increases. If we keep volume constant, then pressure and temperature are directly related. Gay-Lussac’s Law relates pressure and temperature through a math expression: Where P represents pressure and T represents temperature. i and f simply mean before and after the reaction.
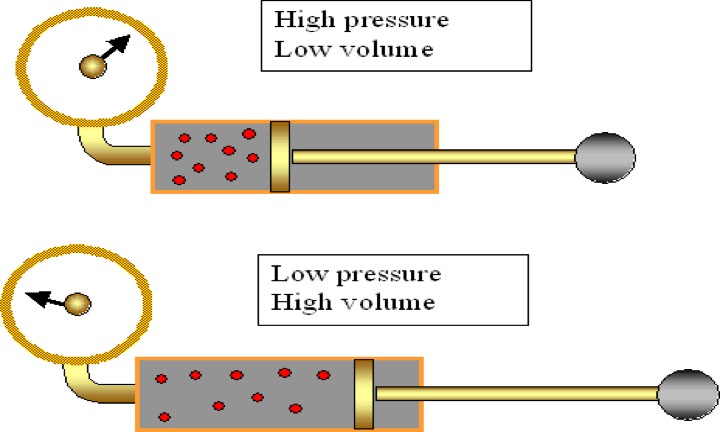
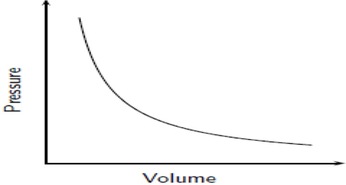
Boyle's Law: If we keep temperature constant, then volume and pressure are inversely related. As one goes up the other goes down If the container gets larger the gas molecules are hitting the walls at a less frequent rate. Therefore, by increasing the volume of a container, we are decreasing the pressure that is felt by that container. The reverse is true if we decrease the volume of a container. Boyle’s Law states that pressure and volume are inversely proportional and they can be described by the equation: Charles Law:
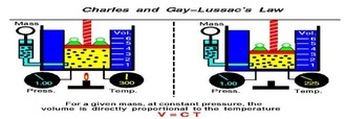
Boyle’s Law looked at the relationship between volume and pressure. Gay-Lussac’s Law looked at the relationship between pressure and temperature Charles Law focuses on the relationship between temperature and volume. As you heat up gas molecules they move quicker and this causes them to hit the sides of the container with greater frequency. If the container is capable of expanding, so that pressure can remain constant, then the volume will increase as the temperature increases. Charles Law can be described by the equation: |
|
|
|
Your Objectives for this Lab
In this lab, you will be using a syringe to design an experiment that studies Charles, Boyle's, and Gay-Lussac's Law. To get a detailed overview of what you will be provided with for this lab and an introduction to each of the concepts, please click on the link below.
Gas Law Lab In the first lab, you will be studying Boyle's Law. You will be provided with a syringe that you will use to study how pressure changes as you change volume. You will start the syringe at a known volume (as measured by the side of the syringe) and a known pressure (1 atm). You will then add weights on top of the syringe in order to increase the pressure. You will observe the change in volume that occurs as a result of these weights. From this change in volume (and initial conditions already stated) you will be able to calculate the new pressure.
In the second lab, you will be studying Charles' Law through the observation of how temperature impacts the volume of a gas. You will have the same syringe and you will now have a water bath. You will change the temperature of this water bath in order to observe how the volume in the syringe changes. You will have access to a hot plate, ice, and dry ice in order to get a wide range of temperatures.
|
Proudly powered by Weebly