- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 1 - Flashing Lights: Concentration Determination
|
Spectrophotometry is a commonly used methodology whereby concentrations of solutions can be determined based upon the amount of color they absorb and/or reflect. Spectrophotometers are extremely expensive and often cost thousands of dollars to purchase even one. Luckily, we can utilize these principles to determine the concentration of unknown solutions.
|
|
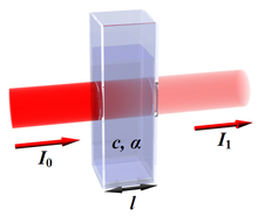
Spctrophotometry is the measure of how much light is absorbed/transmitted through a certain substance. Through these measurements, the concentration of a solution can be determined.
The amount of light that a substance absorbs is directly related to the concentration of a solution. This was summarized through Beer's Law. Beer's Law states that: A = abc
A = absorbance
a = Molar extinction coefficient (a constant for the solution that you are using) b = path length (the distance the light travels in the spectrophotometer) c = concentration of the solution Scientists usually measure absorbance (A) and use this to determine the concentration of a solution that they are working with. The general principle is that as the absorbance increases, so to does the concentration. This is extremely accurate means by which to measure a solutions concentration, but spectrophotometers are extremely expensive and most places cannot afford this machinery. |
|
Today's Lab
|
Please click on the following links to assist you in designing your experiment:
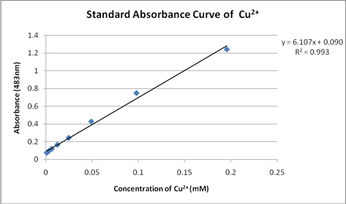
Lab Introduction Part I Lab Introduction Part II While we will not be able to use these fancy instruments, we can utilize the relationship between concentration and absorbance to study solutions. We can study this by utilizing a standard curve. A standard curve is used by plotting known values of a substance (concentration, volume, mass, etc.) and drawing a line of best fit. Once this curve has been generated, then you can use data from your unknown to help determine the value. For example, if you knew the absorbance of 4 solutions and their corresponding concentrations, then you could plot concentration against absorbance. If you then had a solution where you only knew the absorbance, then you could find where that value fell along the best fit line and determine the concentration for this solution. For this lab, you will have copper sulfate pentahydrate (CuSO4), water, a balance, and test tubes. You will not have a specrophotometer and you will not be able to measure the absorbance for this lab. Keep in mind that standard curves can contain other things then what has been described here (e.g. mass vs. volume, mass vs. percent by mass, etc.). For additional information about the background (and helpful hints about your design), please click on the link below. Information for Spectrophotometry Lab |
Proudly powered by Weebly