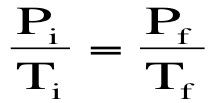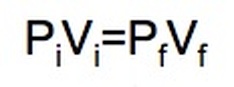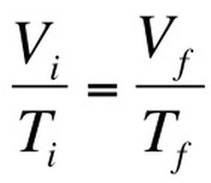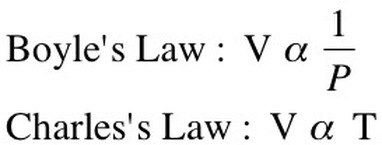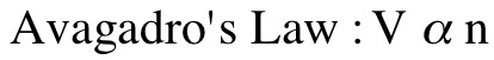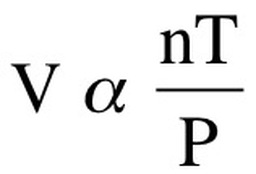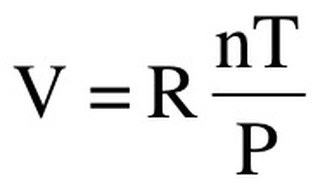- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 9 - Double Bubble, Toil, and the Ideal Gas Law!
|
The ideal gas law is truly a wonderful equation. Through Boyle's, Charles', and Gay-Lussac's Law, we have been able to develop an equation that describes how all three of these laws relate to one another when we take moles of gas into consideration. In this lab, you will be examining the ideal gas law and using it to calculate the number of moles of gas in an unknown liquid.
|
|
We can describe how a gas will change based on the volume, pressure, temperature, and moles of the gas. The gas laws focus on these properties and how changing one affects the other three.
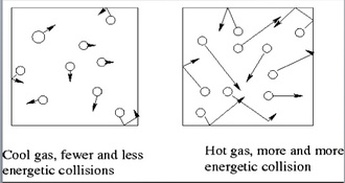
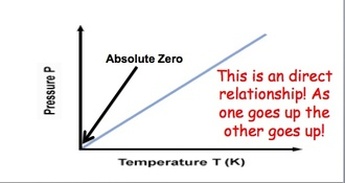
Gay Lussac's Law: Gas molecules move quicker the more we heat them up and increase the temperature. This means that they will be hitting the sides of the container more often and the pressure will also increase as the temperature increases. If we keep volume constant, then pressure and temperature are directly related. Gay-Lussac’s Law relates pressure and temperature through a math expression: Where P represents pressure and T represents temperature. i and f simply mean before and after the reaction.
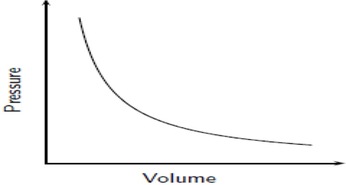
Boyle's Law: If we keep temperature constant, then volume and pressure are inversely related. As one goes up the other goes down If the container gets larger the gas molecules are hitting the walls at a less frequent rate. Therefore, by increasing the volume of a container, we are decreasing the pressure that is felt by that container. The reverse is true if we decrease the volume of a container. Boyle’s Law states that pressure and volume are inversely proportional and they can be described by the equation: Charles Law:
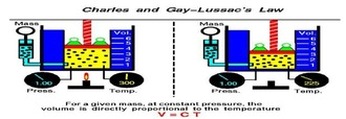
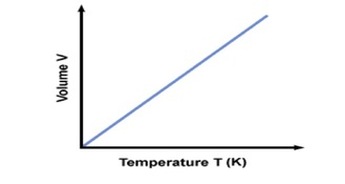
Boyle’s Law looked at the relationship between volume and pressure. Gay-Lussac’s Law looked at the relationship between pressure and temperature Charles Law focuses on the relationship between temperature and volume. As you heat up gas molecules they move quicker and this causes them to hit the sides of the container with greater frequency. If the container is capable of expanding, so that pressure can remain constant, then the volume will increase as the temperature increases. Charles Law can be described by the equation: |
|
The ideal gas law combines Boyle’s, Charles , and Gay-Lussac’s Law into one single equation. We know that as we increase temperature, both pressure and volume will also increase. Additionally, we know that pressure and volume are inversely related. Therefore, we can say that:
The alpha symbol () means proportional to. This just describes if it is a direct (both go up or down at the same time) or indirect (one goes up, then other goes down). Another law that we did not talk about is Avagadro's Law. This law relates the volume of a gas to the number of moles of a gas (n). We say that this is a direct relation and that n is proportional to V:
We can now combine all of these equations to say:
In order to remove the proportional sign, we need a constant that we can multiply one side by in order to make the two sides equal. This is where we insert the gas law constant.
We can rearrange this to get the final equation for the Ideal Gas Law:
PV=nRT
Where P=pressure, V=volume in L, n=moles, R=Gas constant, and T=temperature in Kelvin.
An ideal gas is an imaginary gas whose particles are infinitely small and do not interact with each other. The law works best at room temperature and atmospheric pressure
PV=nRT
Where P=pressure, V=volume in L, n=moles, R=Gas constant, and T=temperature in Kelvin.
An ideal gas is an imaginary gas whose particles are infinitely small and do not interact with each other. The law works best at room temperature and atmospheric pressure
Today's Lab
In this lab, we'll use the ideal gas law to determine the actual percentage of acetic acid in a sample of vinegar. Most vinegars are usually about 5% acetic acid. In order to determine the mass percentage, you will need to react the vinegar with sodium hydrogen carbonate (otherwise known as baking soda). Baking soda and vinegar react by the reaction:
As you can see, carbon dioxide gas (CO2) is produced when you react baking soda and vinegar. Therefore, if we can determine the volume, pressure, and temperature of carbon dioxide produced experimentally, then we can calculate the number of moles of carbon dioxide that we have. Once the moles of carbon dioxide are known, then we can use stoichiometry to calculate the number of moles (and number of grams of vinegar that we started the reaction with.
For a complete list of materials provided and additional background. Please click on the links below:
Ideal Gas Law I
Ideal Gas Law II
Ideal Gas Law III
You will need to develop an apparatus whereby you can capture the carbon dioxide that you are producing. This will allow you to determine the volume of gas that you are producing. You can assume that the pressure of the gas is 1 atm (atmospheric pressure) and you can run this reaction in a water bath in order to keep the temperature stable. HINT - think back to the lab that we did at the beginning of the year to aid you in designing this experiment.
For a complete list of materials provided and additional background. Please click on the links below:
Ideal Gas Law I
Ideal Gas Law II
Ideal Gas Law III
You will need to develop an apparatus whereby you can capture the carbon dioxide that you are producing. This will allow you to determine the volume of gas that you are producing. You can assume that the pressure of the gas is 1 atm (atmospheric pressure) and you can run this reaction in a water bath in order to keep the temperature stable. HINT - think back to the lab that we did at the beginning of the year to aid you in designing this experiment.
Proudly powered by Weebly