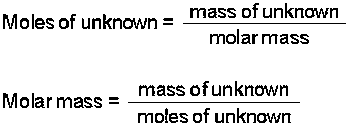- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 2 - Ice Cream Chemistry
|
There are many chemical properties that have been utilized by humans in order to suit our own needs. One of these properties that we have many uses for is a property known as colligative properties. We have used colligative properties to manipulate many things and it is because of these properties that we are able to lower the temperature of a solution so that we can make ice cream. In this lab, you will be studying how a basic property of solutions allows for the lowering of a freezing point and the raising of a boiling point.
|
|
|
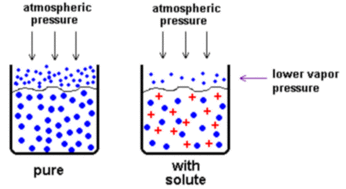
Colligative properties are those properties of solutions that depend on the number of dissolved particles in solution, but not on the identities of the solute. This means that these properties depend only on the amount of solute you have and NOT the identity of the solute that you are adding. The two properties that are most often effected by the identity of the solute are the (1) boiling point and (2) freezing point.
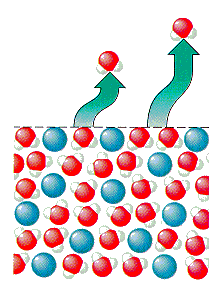
When you add solute to a solution you both raise the temperature at which it boils and lower the temperature at which the solution freezes. The freezing point is lowered because you have added more solute molecules to the solution. More solute molecules means that water molecules have a harder time locating one another, therefore, forming the solid arrangement becomes harder because of the solutes blocking the formation of this structure. This results in a lower freezing point for the solution when you add solute molecules. The boiling point of a solution is increased when you add solute molecules due to the increased amount of solute molecules. When you have more molecules in solution, then you have more molecules blocking the ability of the water molecules from escaping the solution and boiling. This means that a higher temperature is required in order to move water molecules from the liquid phase to the gas phase (i.e. boiling). |
Calculating Change in Freezing Point:
One can calculate the change in freezing point (ΔTf ) relative to the pure solvent using the equation:
ΔTf = iKfm
Kf = the freezing point depression constant for the solvent
m = the molality of the solution or the number of moles of solute in solution per kilogram of solvent
i = the number of ions present per formula unit (e.g., i = 2 for NaCl because the compound split into Na and Cl, so there are two ions present).
Calculating Change in Boiling Point:
The formula used to calculate the change in boiling point (ΔTb ) relative to the pure solvent is similar to that used for freezing point depression:
ΔTb = iKbm
Kb = the boiling point elevation constant for the solvent
m = the molality of the solution or the number of moles of solute in solution per kilogram of solvent
i = the number of ions present per formula unit (e.g., i = 2 for NaCl because the compound split into Na and Cl, so there are two ions present).
Molality:
You will notice that each of these equations have you using the molality of the solution. NOTE - this is molality, not molarity. While both terms describe the concentration of the solution, the molality can be calculated by:
One can calculate the change in freezing point (ΔTf ) relative to the pure solvent using the equation:
ΔTf = iKfm
Kf = the freezing point depression constant for the solvent
m = the molality of the solution or the number of moles of solute in solution per kilogram of solvent
i = the number of ions present per formula unit (e.g., i = 2 for NaCl because the compound split into Na and Cl, so there are two ions present).
Calculating Change in Boiling Point:
The formula used to calculate the change in boiling point (ΔTb ) relative to the pure solvent is similar to that used for freezing point depression:
ΔTb = iKbm
Kb = the boiling point elevation constant for the solvent
m = the molality of the solution or the number of moles of solute in solution per kilogram of solvent
i = the number of ions present per formula unit (e.g., i = 2 for NaCl because the compound split into Na and Cl, so there are two ions present).
Molality:
You will notice that each of these equations have you using the molality of the solution. NOTE - this is molality, not molarity. While both terms describe the concentration of the solution, the molality can be calculated by:
|
Determining Molar Mass Using Colligative Properties:
We can calculate the molar mass of a solute from the measurement of freezing point depression and boiling point elevation. If you can measure the amount of substance initially and the change that it causes in the boiling point, then you will be able to calculate the molality because this will be the only variable remaining. Recall that molality is calculated by: Therefore, if you know the change in boiling point or freezing point, then you can solve for the molality of the solution. Once molality is known, then you can determine the moles of solute that you have:
Since you know the amount of solvent that you used, you can solve for the amount of moles of solute. If you then know how much solute you started with, then you can determine the molar mass through the following equations.
|
Please click on the following handouts to determine the materials that will be available to you for this lab:
Ice Cream Chemistry Part I
Ice Cream Chemistry Part II
Ice Cream Chemistry Part III
Ice Cream Chemistry Part IV
Ice Cream Chemistry Part V
In this lab you will have sodium chloride (NaCl), sucrose (table sugar), water, and a heat source. You will be measuring the change in boiling point and using this change to determine the molar mass of each of these solutions. This lab procedure should be broken up into 3 parts:
Textbook Readings:
For additional help on colligative properties, please see pages 448 - 451 of your textbook.
Ice Cream Chemistry Part I
Ice Cream Chemistry Part II
Ice Cream Chemistry Part III
Ice Cream Chemistry Part IV
Ice Cream Chemistry Part V
In this lab you will have sodium chloride (NaCl), sucrose (table sugar), water, and a heat source. You will be measuring the change in boiling point and using this change to determine the molar mass of each of these solutions. This lab procedure should be broken up into 3 parts:
- Determining the boiling point and freezing point of water (so as to have a reference point)
- Making solutions of salt water and sugar water and determining how the boiling point has changed
- Determining the freezing point of the salt water and sugar water solutions from part 2.
- EXTRA CREDIT - Once you complete this experiment: research, design, and implement the way in which colligative properties can be used to make ice cream. For additional background information about this lab, please click on the link below.
Textbook Readings:
For additional help on colligative properties, please see pages 448 - 451 of your textbook.
Proudly powered by Weebly