- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 4 - Master of Titrations
|
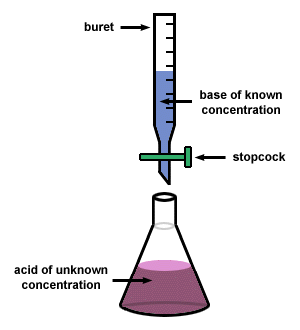
A titration is a procedure whereby the concentration of a solution is determined through the controlled addition of another solution. Titrations are incredibly useful and are utilized in acid/base chemistry quite often. Therefore, becoming a master at these procedures is essential if we are going to good at acid/base chemistry. The lab you will be performing here will allow for you to become the master of titrations.
|
Titration Background Information
|
Often times, we have solutions of acids and/or bases that we do not know the concentration of. A titration is the controlled addition of solution of known concentration/volume to a solution of known volume, but unknown concentration. We use titrations to determine the concentration of an unknown acid or base.
A titration stops once the equivalence point is reached. The equivalence point is the point at which the two solutions used in a titration are present in equivalent (the same) amounts. We use indicators to tell us where the equivalence point is. For a strong acid/strong base reaction, the equivalence point is always at a pH of 7. When we are at the equivalence point, we know that the Moles of acid = moles of base. |
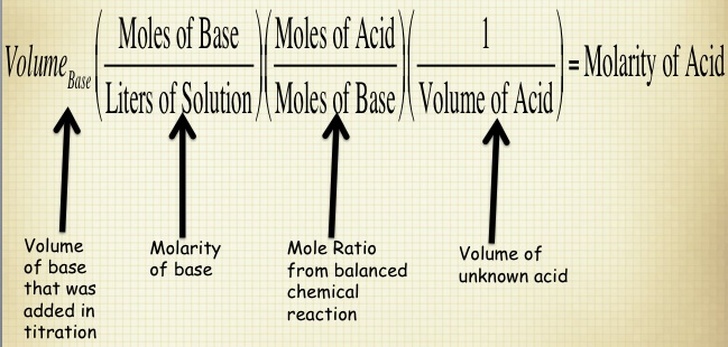
When you are performing a titration, you known the volume of substance that is being delivered by the buret and the concentration of the solution in the buret. You also know the concentration of the substance that you are putting in the flask below the buret. Therefore, if you know all of these components, then you calculate the molarity of your substance by using the stoichiometry shown below. NOTE - this is for determining the concentration of an unknown acid, but you can use this process for ANY substance.
Lab Introduction
When setting up a standardization lab, you need to make sure you are:
(1) Delivering an accurate amount of unknown acid/base to the flask
(2) Putting an acid/base of known concentration into the buret so that the volume is also known.
(3) Titrating until the endpoint is reached so that the concentration of the solution in the flask can be determined.
|
For an introduction to the lab and the materials that you will be provided, please click on the links below:
Titrations Part I Titrations Part II Titrations Part III Titrations Part IV Titrations Part V This lab will focus on the standardization of a solution of hydrochloric acid. Often, when solutions are made, the concentration is unknown. Therefore, the concentration needs to be determined through the process known as standardizing a solution. To standardize an unknown solution, you react that solution with another solution whose concentration is already known. In this lab you will have a solution of HCl that you need to determine the concentration of. If we know the volume of HCl used and the amount of base titrated in and the concentration of that base, then you can determine the concentration of the HCl. The standard solution that you will be using is a solution of sodium carbonate (Na2CO3). You will need to make up a solution of Na2CO3 and then you will need to use this solution to determine the concentration of the solution. You will have HCl, Na2CO3, a buret, and phenolphthalein indicator. NOTE - You will need to be very precise in the making of the sodium carbonate solution! Think about how you can very accurately make a solution so that we know the concentration of the solution. |
Proudly powered by Weebly