- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 5 - Eat Your Vitamins!
|
Vitamins are essential to human life. Without vitamins many essential enzymes in our bodies would not work and, thus, many functions in our bodies would be halted. It is for this reason that many individuals take vitamin supplements. These supplements act to ensure that we get the necessary amount of vitamins in our diet that are required to maintain life. These vitamin supplements advertise the amount of each vitamin that we are obtaining when we take their pills, but how do we know that this is the correct amount? In this experiment, you will be designing and running a lab whereby you will determine if the advertised amount of vitamin C in a supplement aligns with the experimental data that you obtain.
|
Why We Need Vitamin C:
You may have been told that you need to take Vitamin C when you have a cold or are feeling a bit sick, but why specifically do we need Vitamin C in our bodies? The importance of Vitamin C was first noticed long ago when sailors developed scurvy. Watch the video below to learn some astounding facts about Vitamin C.
You may have been told that you need to take Vitamin C when you have a cold or are feeling a bit sick, but why specifically do we need Vitamin C in our bodies? The importance of Vitamin C was first noticed long ago when sailors developed scurvy. Watch the video below to learn some astounding facts about Vitamin C.
|
A vitamin is any compound is any compound that is essential in tiny amount by an organism. These compounds cannot be produced by the organism, so they must be consumed in order to acquire the necessary amounts.
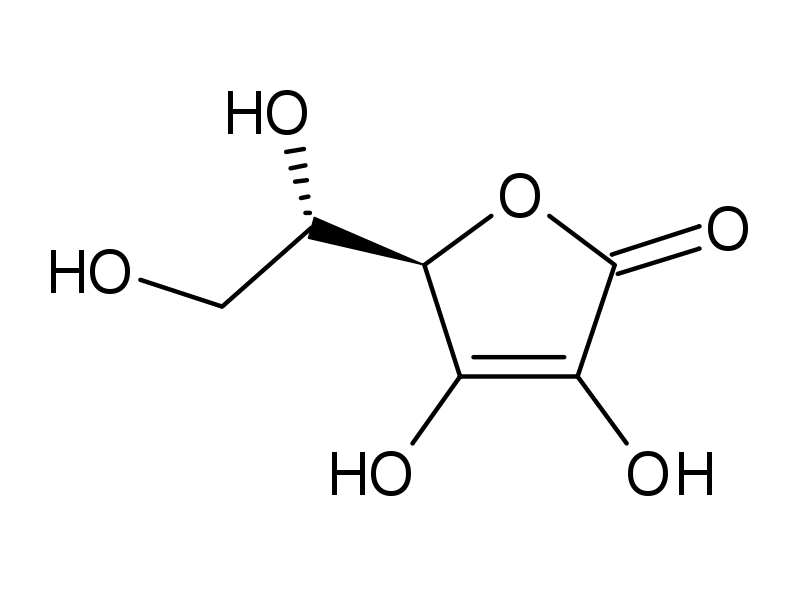
As it pertains to function, vitamins have a very diverse function depending on the vitamin that is being studied. Some have hormone-like functions as regulators of mineral metabolism (e.g., vitamin D), or regulators of cell and tissue growth and differentiation (e.g., some forms of vitamin A). Others function as antioxidants (e.g., vitamin E and sometimes vitamin C).The largest number of vitamins (e.g., B complex vitamins) function as precursors for enzyme cofactors, that help enzymes in their work as catalysts in metabolism. Essentially, most vitamins function as molecules that aid in the function of enzymes. Vitamin C (or ascorbic acid) is used for many functions in the human body. It is used as a cofactor in enzymes (something that helps enzymes work) and as a substance that aids in the synthesis of collagen. It is this synthesis of collagen that is stopped when a person develops scurvy. You may have often heard that you should take vitamin C when you are sick. Vitamin C is found in high concentrations in immune cells, and is consumed quickly during infections. This is the reason why it has been hypothesized as being important in the battle against certain infections. |
Vitamin C Titration Background
|
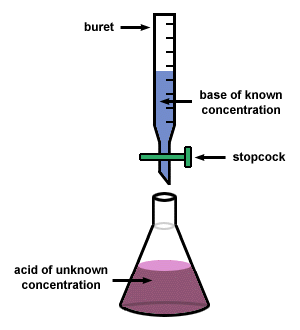
Often times, we have solutions of acids and/or bases that we do not know the concentration of. A titration is the controlled addition of solution of known concentration/volume to a solution of known volume, but unknown concentration. We use titrations to determine the concentration of an unknown acid or base.
A titration stops once the equivalence point is reached. The equivalence point is the point at which the two solutions used in a titration are present in equivalent (the same) amounts. We use indicators to tell us where the equivalence point is. For a strong acid/strong base reaction, the equivalence point is always at a pH of 7. When we are at the equivalence point, we know that the Moles of acid = moles of base. |
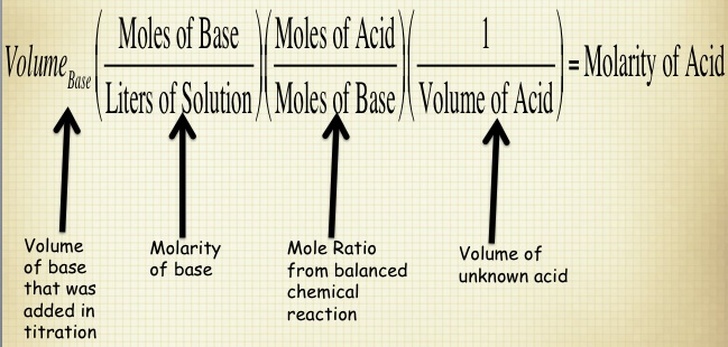
When you are performing a titration, you known the volume of substance that is being delivered by the buret and the concentration of the solution in the buret. You also know the concentration of the substance that you are putting in the flask below the buret. Therefore, if you know all of these components, then you calculate the molarity of your substance by using the stoichiometry shown below. NOTE - this is for determining the concentration of an unknown acid, but you can use this process for ANY substance.
Lab Introduction
|
Please look through the following handouts for materials and background information for this lab:
Eat Your Vitamins Part I Eat Your Vitamins Part II Eat Your Vitamins Part III Many individuals take vitamin C tablets in order to obtain their daily dose of vitamin c. In this lab, you will be determining if the advertised amount on a tablet of vitamin c aligns with the actual amount that is contained within the tablet. You will do this through an acid/base reaction. Ascorbic acid is the active component in the Vitamin C molecule. As you might guess, this is an acid. When you react this with a base you get a typical neutralization reaction: Therefore, if you know the concentration of the sodium hydroxide solution (NaOH), the volume of NaOH that you begin with, and the volume of ascorbic acid that you have, then you can determine the concentration of the ascorbic acid pill.
In this lab, you will have access to burets, Vitamin C tablets, NaOH, and any necessary glassware. You will need to design a lab that addresses the question of how to determine the amount of ascorbic acid in a typical Vitamin C tablet. For more background information and a list of all the materials you will be provided, please click on this link. |
Proudly powered by Weebly