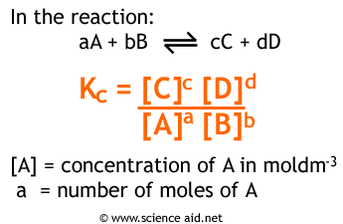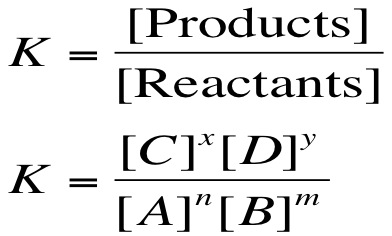- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 7 - Crazy for Le Chatlier
|
We all love equilibriums and Le Chatelier's Principle, but how do we experimentally test this variable? In this lab you will be learning about equilibrium constants and how these can be used to predict how an equilibrium will shift if we alter something on the product or reactants side. You will be designing a lab that allows you to analyze equilibrium constants and how these can be used to describe reactions that have achieved dynamic equilibrium.
|
Le Chatelier's Principle
|
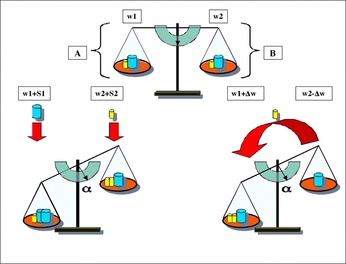
Le Chatelier’s principle states that if we alter something that is on the products or reactants side of an equilibrium, then the system will respond in order to re-establish equilibrium. This principle applies to all reactions in equilibrium.
Change in Concentration: Reactions like to remain at equilibrium. Therefore, if we add more of a product/reactant, then our reaction will shift to the other side in order to compensate for this new amount. Change in Temperature: When a reaction is endothermic, heat is listed as a reactant. When a reaction is exothermic, heat is listed as a product. Therefore we can treat heat like a reactant or a product depending where it is listed. This allows us to know which side heat is added/removed from and, thus, we can determine which way the equilibrium will shift. Change in Pressure: For a gas reaction, changes in pressure can alter the side of the reaction that is favored. If we increase pressure, we increase the concentration of all substances, so the reaction will shift to the side that produces less molecules If we decrease pressure, we decrease the concentration of all substances, so the reaction will shift to the side that produces more molecules |
Equilibrium Constants
After equilibrium is attained, the concentration of products and reactants does not change. Therefore, the ratio of these concentration also remains constant. While the amounts of each product/reactant may change, the ratio will not change.
Since the ratio of products and reactants is not changing, we can calculate the equilibrium constant (Keq) for a given equilibrium. For the equilibrium nA + mB ⇌ xC + yD The equilibrium constant is calculated by: |
There are many different types of equilibrium that exist. Often, a solid compound is in equilibrium with ions. This results in a chemical formula that often looks like:
AgCl (s) ⇌ Ag (aq) + Cl (aq)
This means that solid silver chloride can be in equilibrium with its ions when you dissolve it in solution. This equilibrium can be described using the equilibrium constant known as a solubility product (Ksp). This lab will require some extensive background information in order for you to design it. Therefore, please click on the handout below to get a full overview of the focus of this lab and the materials that will be provided to you:
Le Chatelier Part I
Le Chatelier Part II
Le Chatelier Part III
You will be accomplishing two things during this lab:
1. Determining the Ksp of sodium chloride through evaporation.
2. Determining the Ksp of sodium hydrogen tartrate through the utilization of a titration.
Le Chatelier Part I
Le Chatelier Part II
Le Chatelier Part III
You will be accomplishing two things during this lab:
1. Determining the Ksp of sodium chloride through evaporation.
2. Determining the Ksp of sodium hydrogen tartrate through the utilization of a titration.
Proudly powered by Weebly