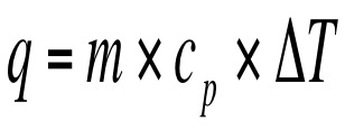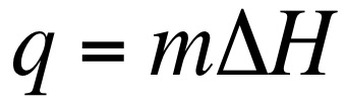- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 10 - Catching Fire
|
In this lab, you will be utilizing the principles of calorimetry to determine the specific heat of a metal and the enthalpy of fusion of ice. The specific heat and enthalpy of fusion were considered constants and were values that were given, but where do these values come from originally? These values can only be determined through controlled experiments. By using your basic knowledge of thermochemistry, you will be able to determine values that are often given to you.
|
Specific Heat and Latent Heat of Substances
|
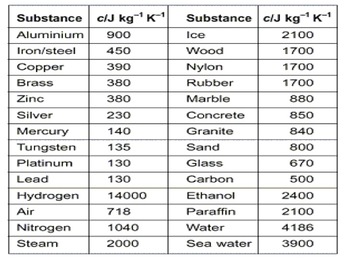
The amount of energy that is transferred to a material is dependent on the nature of the material receiving the energy. We can quantify the amount of heat transferred using the specific heat. The specific heat is the amount of energy required to raise the temperature of one gram of substance one degree Celsius. The specific heat can be represented by the symbol Cp.
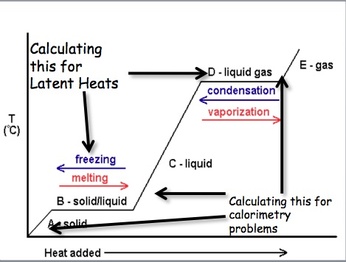
We can measure the amount of heat that is released or absorbed during a chemical process. The process of measuring this change is known as calorimetry. Calorimetry is the science of measuring the heat of a chemical reaction. In the equation q represents the heat gained/released, Cp is the specific heat, and ΔT = Final Temperature – Initial Temp. On a heating curve, these are the areas of positive slope.
We can also calculate the amount of energy that is necessary to convert a substance from one phase to another. This is known as calculating the latent heat. The latent heat is the energy absorbed as a substance changes phases. In the above equation m = mass of compound and ΔH = Enthalpy values for phase changes (constants). On a heating curve these are the values for the areas of no slope.
|
|
In this lab you will be calculating two values:
(1) The heat of fusion (ΔHfusion) of ice (2) The specific heat of a metal For a list of the materials that you will be provided and additional background information, click on the links below: Catching Fire Part I Catching Fire Part II Catching Fire Part III Catching Fire Part IV In the first lab that you design, you will be determining the heat of fusion for ice. Recall, if you add heat to a solution going through a phase change, then the temperature does not change until all of the substance has been converted into the new phase. This energy that is added is the energy that is required for all the atoms to break the forces that are attracting them to other molecules in that phase. This additional energy to change a solid to a liquid is known as the heat of fusion (ΔHfusion). In the first lab, you will determining the ΔHfusion for ice as it goes from a solid to a liquid. Since this lab requires a greater amount of chemistry knowledge, please look at the procedure linked here and create a new procedure that has clear modifications. In the second lab, you will be determining the specific heat of a metal. Please read the introduction to this lab linked below and develop a procedure based on the information and materials that are provided. Specific Heat Lab Introduction |
Extra Resources:
Proudly powered by Weebly