- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 3 - Battery Chemistry
|
Batteries are powered by a very simple reaction known as a redox reaction. This is simply a process by which we use the process of transferring electrons in order to generate products. These types of reactions characterize a large amount of reactions that occur all around us (and within our own bodies!). In these labs you will be studying the basics of redox reactions and you may just develop enough skills to start making your own batteries.
|
Introduction to Redox Reactions
|
Redox Reactions:
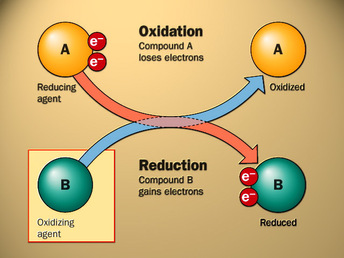
Redox reactions or oxidation - reduction reactions are all reactions that involve the transfer of electrons from one element/compound to another element/compound. During these reactions, substances are either oxidized or reduced. When something loses electrons it is oxidized. When something gains electrons it is reduced. A quick mnemonic device to aid in this understanding is: OIL RIG
Oxidized
Is Losing Reduction Is Gaining Therefore, for redox reactions, you can identify the substance that is being reduced as the one that is gaining electrons and becoming more negative. You can identify the substance that is being oxidized as the one that is losing electrons and, thus, is becoming more positive. |
Assigning Oxidation States
|
When looking at a chemical reaction, you can identify which chemicals are being oxidized and which are being reduced by the process of assigning of oxidation states to each element/compound. You assign oxidation states to an element based on the following rules:
1. An element that is not combined with any other element (H2, Br2, Na, etc) has a oxidation state of 0. 2. Ions have an oxidation state that is the same as their charge (e.g.Na-1 = -1, Cl- = -1, O2-= -2, etc). 3. The oxidation state of oxygen is -2, except for H2O2 where it is -1. 4.The oxidation state of hydrogen is always +1, except when bound the metals (LiH, NaH, etc.). Then its state is +1. 5. Florine always has an oxidation number of -1. Other halogens usually occur as negative charges except when bound to oxygen. 6. In a neutral molecule the sum of all the oxidation states on each individual atom must be. In charged compounds, the sum of all the oxidation states must equal that charge. |
Introduction to Lab
|
Please click on the links below for a list of materials and introduction materials that you have for this lab:
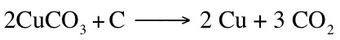
Making a Battery Part I Making a Battery Part II Making a Battery Part III There are many common redox reactions that can be studied in the laboratory. The first reaction that will be studied is the reaction between copper carbonate (CuCO3) and carbon (C). This reaction can be described by the equation: This reaction was discovered thousands of years ago by our ancestors. The discovery of the reaction was purely accidental. Thousands of years ago our ancestors were burning pieces of wood (which are made mostly of carbon) and thy used pieces of copper carbonate as the stone in the circle. As the fire burned and copper carbonate reacted with carbon, the supposed stones that they thought they had used turned into a shiny puddle of metallic copper. This lead to the discovery of smelting (the process of obtaining metals from copper ore) and the end of the Stone Age.
Using copper carbonate, charcoal (a molecule of only carbon, and other tools at your disposal, you will redesign this original reaction. |
|
The second experiment that you will be designing involves the observation of numerous oxidation states that are present for manganese (Mn). Many elements exist in multiple oxidation states and this change in oxidation state. The permaganate ion (MnO4) can have different colors based on the oxidation state and the acidity of the solution. The colors, oxidation states, and ion are seen in the table below.
You can produce these different colors by reacting sodium bisulfite (NaHSO3) with potassium permanganate (KMnO4) in acidic, basic, and neutral solutions. You will be designing an experiment where you can observe the colors that form when this reaction is carried out in acidic, basic, and neutral solution. NOTE - You will have NaOH and H2SO4 to aid in your production of acidic, basic, and neutral solutions.
|
Proudly powered by Weebly