- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 6 - Swim with the Fishes
|
We all love going to the ocean, but have you ever stopped and wondered what actually is in the ocean? Yes, we know that it contains salt water, but what kinds of elements can you find lurking in these waters? In this lab, you will be analyzing the different types of salts that make up the salt water in the ocean by Santa Monica, CA. You will be performing experiments to determine if the levels match the typical levels or if they are higher or lower than what is normally seen in these waters.
|
|
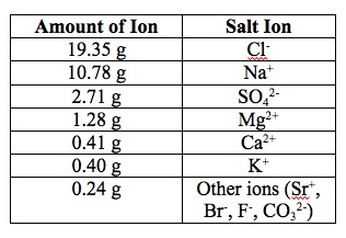
Oceans are complex ecosystems. There is a wide array of animals, plants, and bacteria that reside within the Earth's oceans. Additionally, the ocean waters contain more than just the typical ions you would expect. If you have 1000 g of ocean water, typically 965 g is water and about 35 g are various salts. There are many different type of salts that can be dissolved in the ocean. Of the 35 g that is classified as being salt, the following ions are present:
As you can see, there are many ions that are floating around in the seawater. We can perform experiments to analyze seawater to determine if these numbers are accurate and to determine if there are other ions floating around in the water.
|
|
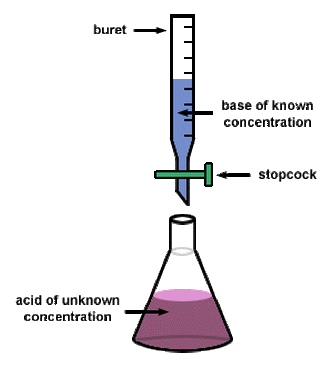
Often times, we have solutions of acids and/or bases that we do not know the concentration of. A titration is the controlled addition of solution of known concentration/volume to a solution of known volume, but unknown concentration. We use titrations to determine the concentration of an unknown acid or base.
A titration stops once the equivalence point is reached. The equivalence point is the point at which the two solutions used in a titration are present in equivalent (the same) amounts. We use indicators to tell us where the equivalence point is. For a strong acid/strong base reaction, the equivalence point is always at a pH of 7. When we are at the equivalence point, we know that the Moles of acid = moles of base. |
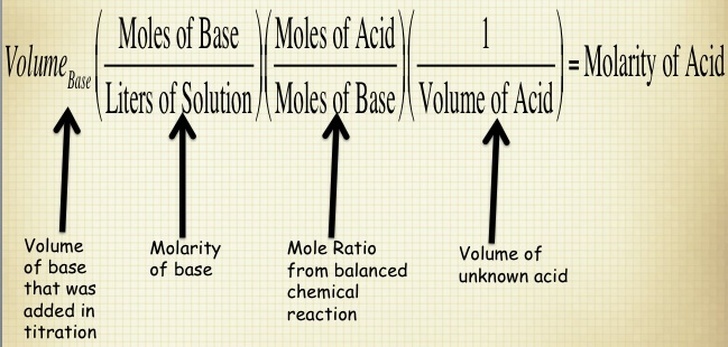
When you are performing a titration, you known the volume of substance that is being delivered by the buret and the concentration of the solution in the buret. You also know the concentration of the substance that you are putting in the flask below the buret. Therefore, if you know all of these components, then you calculate the molarity of your substance by using the stoichiometry shown below. NOTE - this is for determining the concentration of an unknown acid, but you can use this process for ANY substance.
Often times, when certain ions are in solution they combine together to form solids (often called precipitates). Whether a precipitate is formed or not is often used to verify the presence of certain. Whether ions form depend on the following solubility rules:
Using these rules, we can determine if a precipitate (solid) will form when we mix two different types of ions together.
- All common compounds of that form when bound to elements in Group I are soluble (dissolve in water). Additionally, all elements that bind to ammonium ions (NH4) are soluble.
- All compounds that contain nitrates (NO3), acetates (CH3COO), and chlorates (ClO3)are soluble.
- All binary compounds of the halogens (other than F) with metals are soluble, except those bound to Ag, Hg, and Pb. Pb halides are soluble in hot water.
- All elements bound to sulfates (SO4) are soluble, except those bound to barium, strontium, calcium, lead, silver, and mercury.
- Except for rule 1, compounds that contain carbonates (CO3), hydroxides (OH), oxides (O), silicates (SiO4), and phosphates (PO4) are insoluble.
- Compounds that contain sulfur insoluble except for those bound to calcium, barium, strontium, magnesium, sodium, potassium, and ammonium.
Using these rules, we can determine if a precipitate (solid) will form when we mix two different types of ions together.
|
You will design and run an experiment in this lab that tests for the amount of chloride and sulfate ions in a solution. You will test for this using titrations. Since this involves a great deal of extra chemistry knowledge, you will be reading the introduction to the lab in order to aid in your experimental design.
Swim with the Fishes Part I Swim with the Fishes Part II Swim with the Fishes Part III Using this introduction and the information that was provided to you on this webpage, you need to design an experiment to help answer the following issues: 1. What is the density of the seawater sample and the total mass of the dissolved solids? 2. What is the concentration of chloride ions in the seawater? 3. What is the concentration of sulfate ions? This lab will need to be broken up into 3 parts in order to properly designed. |
Proudly powered by Weebly