- Home
- Science Fair Documents
- Lab 1 - Flashing Lights
- Lab 2 - Ice Cream Chemistry
- Lab 3 - Battery Chemistry
- Lab 4 - Master of Titrations
- Lab 5 - Eat Your Vitamins!
- Lab 6 - Swim with the Fishes
- Lab 7 - Crazy for Le Chatlier
- Lab 8 - Direct and Indirect Love of Chemistry
- Lab 9 - Double Bubble, Toil and the Ideal Gas Law!
- Lab 10 - Catching Fire
- Lab 11 - Real Life Water White
- Lab 12 - Real Life CSI
Lab 11 - Real Life Walter White
|
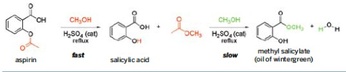
The lab you will be performing here would definitely make Walter White proud. Organic chemistry is the branch of chemistry that studies carbon and all of the reactions that it undergoes. Organic chemistry is a very complex and extensive branch of chemistry that is responsible for many of the chemicals that are currently synthesized for pharmaceuticals, food flavorings and many other substances. In this lab, you will be synthesizing the oil of wintergreen from household aspirin. Oil of wintergreen is the compound that is responsible for the wintergreen smell and taste that is added to many common foods.
|
Functional Groups and Esterification Reactions
|
Functional Groups:
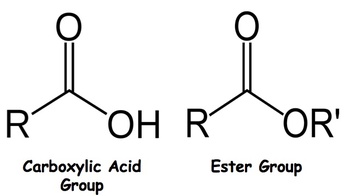
Carbon can undergo many complex reactions. This is due to the fact that it has four valance electrons that can readily bond with many different elements/compounds. This ability to bond results in a wide variety of reactions that is the focus of organic chemistry. Because of its ability to bond with many elements, many common functional groups are seen. A functional group specific groups of atoms or bonds within molecules that are responsible for the characteristic chemical reactions of those molecules. The reaction we will be studying in this lab involves taking a carboxylic acid group and turning it into an ester group. A picture of what carboxylic acid and ester groups look like can be seen in the picture below. NOTE - R represents any carbon group that may continue off of it. When no letters are written, this means carbon is found here. Esterfication:
The process of replacing a carboxylic acid group with an acid is known as esterfication. Because you are replacing the carboxylic acid with the less reactive ester group, this is often a very slow reaction and requires some form of a catalyst in order to go to completion (H2SO4 is often a very good catalyst for these types of reaction). Basically, you substitute the ester group you want to attach for the carboxylic acid group that is already attached. This process of substitution is a common type of reaction that is seen in organic chemistry and it is the best way in which to remove the carboxylic acid group in order to attach the ester group that you want in your final product. Since this is a relatively complex lab, please modify the procedure that is located here. |
|
|
There are two other main concerns that one often encounters during an organic synthesis procedure: separation of products from the reaction mixture and how to determine if you have made the product that you were attempting to make.
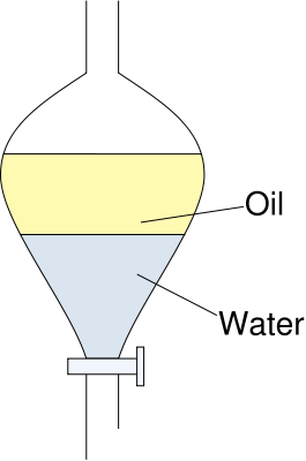
Seperatory Procedures: Once you have completed the reaction, you will be left with a brown liquid in the reaction vessel. This is a solution that contains methanol, crude methyl salicylate, sulfuric acid, unreacted aspirin, and other impurities. Methyl salicylate is soluble in methanol, but not soluble in ice water. This means if you add ice water and allow for the solution to separate, than we can pull out our desired product. For a clear outline of how to do this seperatory procedure, click here. Determination of Purity: Once we have isolated the product, we need to be able to determine the purity of the product that you just produced. How do you provide evidence that supports the idea that you just produced a product? The best way to achieve this is through the determination of the density and freezing point of the product. We know that the density of pure methyl salicylate is 1.1825 g/mL, so the density of the product will give us some idea of its purity. Therefore, if the density of the new substance is close to this value, then we know that we may have produced the desired product. Density is calculated by dividing the mass of the substance by the volume of the substance. Therefore, if we can calculate the mass and measure the volume of a methyl salicylate sample, then we can calculate the density of our new substance. That's not sufficient, however. Even if the density of our product is very close to 1.1825 g/mL, for all we know our product might be a small amount of methyl salicylate mixed with a large amount of some impurity that has the same density. The freezing point our product will give us a better idea of its purity. Pure methyl salicylate freezes at -8.3 C. Therefore, the closer the freezing point is to -8.3 C, the higher the purity of our final compound. You will have a test tube, thermometer, and ice bath in order to measure the freezing point of your substance. |
Today's Lab
|
In this lab you will be taking household aspirin and converting it to methyl salicylate (the wintergreen smell). This reaction can be run to completion with the use of only sulfuric acid and methonal. You know that the reaction works best at a temperature of 60 C an it tends to take 60 minutes at this temperature to get the reaction to run to completion. Additionally, you know that you will get two layers and that you will need to isolate the final product from these two layers.
Please look at the following handouts to let you know the materials that you will be using and additional background information: Real Walter White Part I Real Walter White Part II Real Walter White Part III |
Extra Readings:
Organic Chemistry readings that may aid in your experimental design.
Proudly powered by Weebly